Chemistry Concepts in Action
On-Site School Program
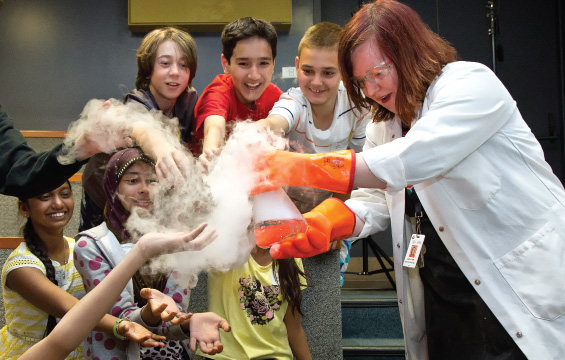
Curriculum Connections:
Strands A and C : STEM Investigation Skills and Chemistry: The Nature of Matter
| A1.2 | apply a scientific experimentation process and associated skills to conduct investigations, making connections between their observations and findings and the scientific concepts they are learning |
|---|---|
| A1.5 | apply their knowledge and understanding of safe practices and procedures, including the Workplace Hazardous Materials Information System (WHMIS), while planning and carrying out hands-on investigations |
| C1.1 | assess social, environmental, and economic impacts of processes associated with the life cycle of consumer products, considering the elements and compounds used to make them, and suggest ways to enhance positive impacts and/or minimize negative impacts |
| C2.5 | investigate the physical and chemical properties of elements, and use their findings to relate these properties to the organization of the periodic table, classify elements, and identify patterns in the periodic table |
| C2.6 | investigate and describe physical and chemical properties of elements and compounds, including those that make up common household products |
| C2.7 | describe the relationship between the structure of simple compounds and their chemical formulas |
Strands A and C : Scientific Investigation Skills and Chemistry: Chemical Reactions
| A1.1 | formulate scientific questions about observed relationships, ideas, problems, and/or issues, make predictions, and/or formulate hypotheses to focus inquiries or research |
|---|---|
| A1.4 | apply knowledge and understanding of safe practices and procedures when planning investigations(e.g.,appropriate techniques for handling, storing,and disposing of laboratory materials [following the Workplace Hazardous Materials Information System–WHMIS];safe operation of optical equipment;safe handling and disposal of biological materials),with the aid of appropriate support materials(e.g.,the Reference Manual on WHMIS website;theLive Safe! Work Smart! website) |
| C2.1 | use appropriate terminology related to chemical reactions, including, but not limited to: compounds, product, and reactant |
| C3.3 | describe the types of evidence that indicate chemical change |
| C3.4 | write word equations and balanced chemical equations for simple chemical reactions |
| C3.5 | describe, on the basis of observation, the reactants in and products of a variety of chemical reactions, including synthesis, decomposition, and displacement reactions |
| C3.7 | describe how the pH scale is used to classify solutions as acidic, basic, or neutral |
Strands A and C : Scientific Investigation Skills and Chemistry: Chemical Reactions and Their Practical Applications
| A1.1 | formulate scientific questions about observed relationships,ideas,problems,and/or issues, make predictions,and/or formulate hypotheses to focus inquiries or research |
|---|---|
| A1.4 | apply knowledge and understanding of safe practices and procedures when planning investigations(e.g.,appropriate techniques for handling, storing,and disposing of laboratory materials [following the Workplace Hazardous Materials Information System–WHMIS];safe operation of optical equipment;safe handling and disposal of biological materials),with the aid of appropriate support materials(e.g.,the Reference Manual on WHMIS website;theLive Safe! Work Smart! website) |
| C1.2 | identify practical applications of chemical reactions in a particular profession, and assess the associated hazards, including hazards associated with the handling and disposal of chemicals |
| C2.1 | use appropriate terminology related to chemical reactions, including, but not limited to: dilute, neutralization, product, reactant, and word equation |
| C3.2 | name and write the formulae for simple ionic and molecular compounds |
| C3.3 | write word equations and balanced chemical equations for simple chemical reactions |
| C3.4 | describe the process of neutralization for simple acid–base reactions |
| C3.5 | describe how the pH scale is used to identify the concentration of acids and bases |
Strands A, B, C, E and F : Scientific Investigation Skills, Matter, Chemical Trends, and Chemical Bonding, Chemical Reactions, Solutions and Solubility and Gases and Atmospheric Chemistry
| A1.1 | formulate relevant scientific questions about observed relationships, ideas, problems, or issues, make informed predictions, and/or formulate educated hypotheses to focus inquiries or research |
|---|---|
| A1.4 | apply knowledge and understanding of safe laboratory practices and procedures when planning investigations by correctly interpreting Workplace Hazardous Materials Information System (WHMIS) symbols; by using appropriate techniques for handling and storing laboratory equipment and materials and disposing of laboratory materials; and by using appropriate personal protection (e.g., wearing safety goggles) |
| B2.1 | use appropriate terminology related to chemical trends and chemical bonding, including, but not limited to: atomic radius, effective nuclear charge, electronegativity, ionization energy, and electron affinity |
| B3.5 | compare and contrast the physical properties of ionic and molecular compounds (e.g., NaCl and CH4 ; NaOH and H2O) |
| C2.1 | use appropriate terminology related to chemical reactions, including, but not limited to: neutralization, precipitate, acidic, and basic |
| C3.1 | identify various types of chemical reactions, including synthesis, decomposition, single displacement, double displacement, and combustion |
| C3.2 | explain the difference between a complete combustion reaction and an incomplete combustion reaction (e.g., complete and incomplete combustion of hydrocarbon fuels) |
| E2.1 | use appropriate terminology related to aqueous solutions and solubility, including, but not limited to: concentration, solubility, precipitate, ionization, dissociation, pH, dilute, solute, and solvent |
| E3.3 | explain the effects of changes in temperature and pressure on the solubility of solids, liquids, and gases (e.g., explain how a change in temperature or atmospheric pressure affects the solubility of oxygen in lake water) |
| F3.2 | describe the different states of matter, and explain their differences in terms of the forces between atoms, molecules, and ions |
| F3.3 | use the kinetic molecular theory to explain the properties and behaviour of gases in terms of types and degrees of molecular motion |
Strands A and C : Scientific Investigation Skills And Career Exploration and Chemicals in Consumer Products
| A1.1 | formulate relevant scientific questions about observed relationships, ideas, problems, or issues, make informed predictions, and/or formulate educated hypotheses to focus inquiries or research |
|---|---|
| A1.4 | apply knowledge and understanding of safe laboratory practices and procedures when planning investigations by correctly interpreting Workplace Hazardous Materials Information System (WHMIS) symbols; by using appropriate techniques for handling and storing laboratory equipment and materials and disposing of laboratory materials, including biological waste (e.g., techniques to prevent contamination of specimens); and by using appropriate personal protection (e.g., wearing gloves when handling biological specimens) |
| C2.1 | use appropriate terminology relating to chemical reactions and chemical products, including, but not limited to: synthesis, decomposition, neutralization, polymerization, combustion, single and double displacement, pH, solvent, organic, inorganic, and dilution |
| C3.1 | describe the types of chemical reactions (e.g., synthesis, single displacement, double displacement, decomposition, combustion, polymerization, neutralization) and the signs of chemical change in each |
| C3.3 | explain the function of the pH scale and how pH test results are interpreted |
Strands A, C, D, E and F : Scientific Investigation Skills, Structure And Properties of Matter, Energy Changes and Rates of Reaction, Chemical Systems and Equilibrium and Electrochemistry
| A1.1 | formulate relevant scientific questions about observed relationships, ideas, problems, or issues, make informed predictions, and/or formulate educated hypotheses to focus inquiries or research |
|---|---|
| A1.4 | apply knowledge and understanding of safe laboratory practices and procedures when planning investigations by correctly interpreting Workplace Hazardous Materials Information System (WHMIS) symbols; by using appropriate techniques for handling and storing laboratory equipment and materials and disposing of laboratory materials; and by using appropriate personal protection (e.g., wearing safety goggles) |
| C2.1 | use appropriate terminology related to structure and properties of matter, including, but not limited to: orbital, emission spectrum, energy level, photon, and dipole |
| D3.1 | compare the energy changes resulting from physical change (e.g., boiling water), chemical reactions (e.g., bleaching a stain), and nuclear reactions (e.g., fission, fusion), in terms of whether energy is released or absorbed |
| E2.2 | predict, applying Le Châtelier’s principle or the reaction quotient for a given reaction, how various factors (e.g., changes in volume, temperature, or concentration of reactants or products in a solution) would affect a chemical system at equilibrium, and conduct an inquiry to test those predictions |
| E3.2 | explain the concept of chemical equilibrium and how it applies to the concentration of reactants and products in a chemical reaction at equilibrium |
| E3.3 | explain Le Châtelier’s principle and how it applies to changes to a chemical reaction at equilibrium |
| F2.2 | conduct an inquiry to analyse, in qualitative terms, an oxidation-reduction (redox) reaction |
| F3.1 | explain redox reactions in terms of the loss and gain of electrons and the associated change in oxidation number |
Strands A and D : Scientific Investigation Skills and Electrochemistry
| A1.1 | formulate relevant scientific questions about observed relationships, ideas, problems, or issues, make informed predictions, and/or formulate educated hypotheses to focus inquiries or research |
|---|---|
| A1.4 | apply knowledge and understanding of safe laboratory practices and procedures when planning investigations by correctly interpreting Workplace Hazardous Materials Information System (WHMIS) symbols; by using appropriate techniques for handling and storing laboratory equipment and materials and disposing of laboratory materials; and by using appropriate personal protection (e.g., wearing safety goggles) |
| D3.1 | explain the concepts of oxidation and reduction in terms of the chemical changes that occur during redox reactions |
Sensory and Safety Considerations
Sensory Considerations: This program features moderate to loud noises; the Presenter will warn students of upcoming loud noises during combustion.
Safety Considerations: Chemical and combustible materials are used in this program. The Ontario Science Centre will outline safety procedures at the beginning of the program and follow them closely. Under teacher supervision and by following the safety instructions, students may be invited to participate in certain demonstrations.


